At InfoBloom, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What Is Hydrated Silica?
Hydrated silica consists of the chemical compound silicon dioxide in a form that is bound to water molecules. The water content of this compound can vary, and if the silicon dioxide is completely dissolved in water, the compound is called silicic acid. In nature, hydrated silica occurs in various forms, for example as diatomaceous earth and as opals. It can also be manufactured, and then usually contains enough water to produce a white, gelatinous substance that lacks both taste and odor, and which can be used as a mild abrasive and to increase the viscosity, or thickness, of various products. This substance is non-toxic, and is commonly used in toothpastes, cosmetics, and body-care products.
Silicon, also known as silica and which has the chemical formula SiO2, is a metalloid, meaning it has some of the characteristics of metals and some of the characteristics of non-metals. It is not to be confused with silicone, which is a polymer. Silicon is one of the most common chemical elements on earth, and in the form of silicon oxide, it is found in sand, quartz, flint, granite, and many other minerals.

In the form of hydrated silica, it is also found in the cell walls of a type of algae called diatoms. Diatomaceous earth, a type of soft, sedimentary rock mined for a variety of industrial uses, consists of hardened sediments of fossilized diatoms. It's also found as opals, a type of gemstone that consists of silicon dioxide with a water content that is commonly 6-10%.

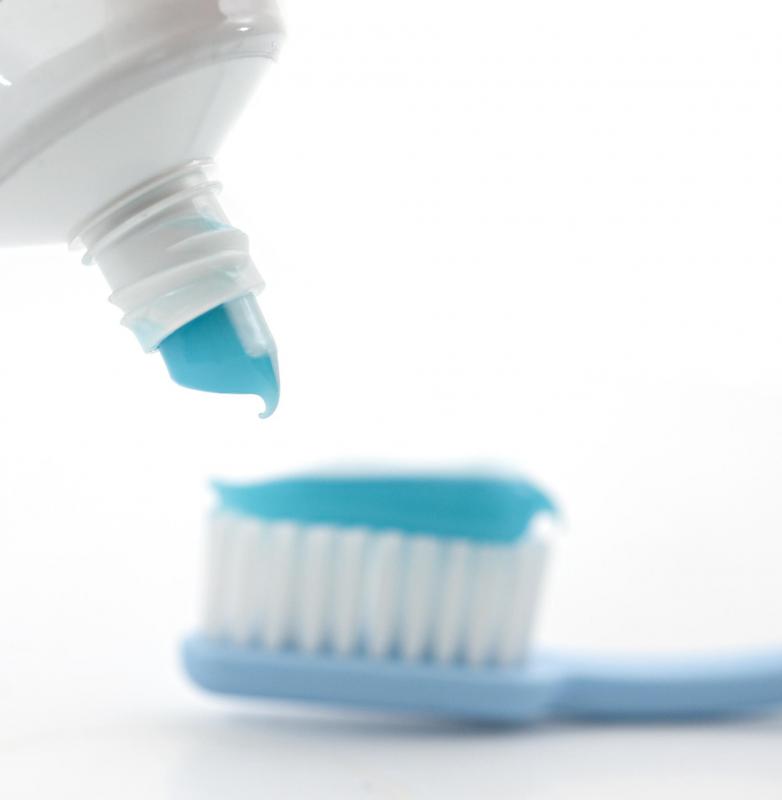
A very common use for manufactured hydrated silica is as a toothpaste ingredient, prized for its abrasive properties which help remove plaque and calculus. In regular toothpaste, the hydrated silica is often milled until the grains are about 8 microns in diameter. For use in special whitening toothpastes, the grains are somewhat larger, commonly 10 microns in diameter, which gives a more abrasive effect. In older types of toothpaste, diatomaceous earth was used for the same purpose. This substance is also used in various personal care products such as skin creams, deodorants, and facial scrubs.

In chemistry, hydrated silica is sometimes referred to as an inert substance, meaning it is not chemically reactive. This substance is not known to be toxic, or cause cancer, and the Food and Drug Administration (FDA) in the United States has classified it as Generally Recognized As Safe (GRAS). It is important not to confuse this type of silica with crystalline silica, which is a toxic substance.
AS FEATURED ON:
AS FEATURED ON:













Discussion Comments
What the FDA says is not always correct afterward. You can't track if a human died of cancer after years of using a toothpaste with some chemicals in it which are approved by the FDA. It's logical that a body can't go on forever. Living food for living people. Not chemicals for living people.
This is a great article and I fully appreciate that this substance is considered safe to be used in personal care products. As it is in so many things that can only be a good thing.
The only thing I would like to say is that hydrated silica in toothpaste isn't always a good thing.It can cause some damage to the surface of your teeth, especially if they are not brushed correctly.
I've been in the dental profession for many years and I know many people struggle with mouth care. So I really recommend that those with gum disease avoid this in their toothpaste products.
Post your comments