At TheHealthBoard, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What is Trypsin?
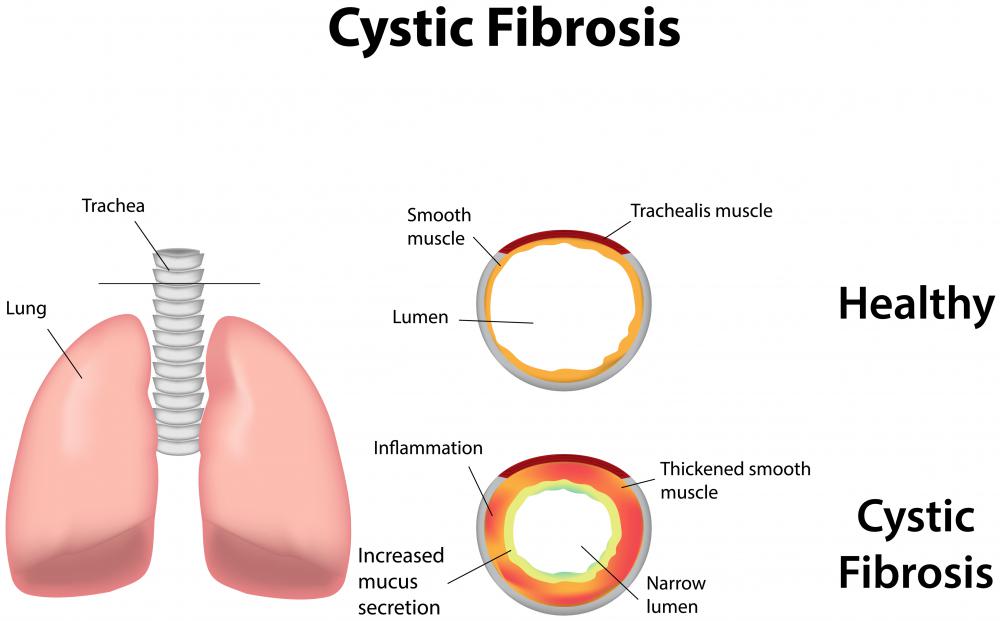
Trypsin is part of the digestive system and degrades proteins, making it an enzyme known as a protease. It is initially formed as a larger, inactive molecule in the pancreas and is transported to the small intestine, where it is activated to digest food molecules. This protease is also a regulator of many other digestive proteases. A lack of its production in the pancreas is a component of the disorder cystic fibrosis.
Food that is made up of proteins is partially broken down in the stomach by proteases, and the smaller pieces travel into the small intestine. Proteases are enzymes that speed up the breakdown of proteins. Amino acids are the building blocks of proteins, and they are linked together by peptide bonds. The end goal of the digestion of proteins is to degrade them to amino acids, which can be utilized in cellular metabolism.

Trypsin is originally synthesized in the pancreas in a form known as trypsinogen, a zymogen. This larger molecule is inactive until it is transported to the small intestine and acted upon by enteropeptidase, another type of protease. Once this step has taken place, trypsin can activate itself. In addition to degrading proteins in digestion, this substance activates a number of other zymogens. Because of its important role in activating so many other proteases, pancreatic juice contains an inhibitor that blocks its activity while it is in the pancreas.

Along with chymotrypsin and a number of other proteases, trypsin is in a class known as serine proteases. This class of proteases is comprised of endopeptidases. They cleave within a protein molecule, instead of nibbling away at the ends. Serine proteases have a key serine amino acid at their active site.
Chymotrypsin and trypsin are very similar to each other in structure and chemical composition. The main difference is in the substrates they prefer. Trypsin cleaves by the amino acids arginine and lysine, while chymotrypsin prefers tryptophan, tyrosine, and phenylalanine. They are both optimal in the neutral pH environment of the small intestine.

A lack of transport of trypsin, and other digestive enzymes from the pancreas, is one of the factors involved in cystic fibrosis. This causes obstructions in the intestines to build up, interfering with the absorption of nutrients. Newborn babies are usually tested for the presence of this enzyme in their blood, and a high level is typically indicative of cystic fibrosis. For older individuals, activity in stool can be measured. If the results are negative, further testing is merited.
AS FEATURED ON:
AS FEATURED ON:














Discussion Comments
What is the pH of trypsin?
The pancreas is one of those things that everyone knows is important but no one really knows what it does. I remember having a high school biology teacher who told us to think of the pancreas as the laboratory of the body. It produces various enzymes and other vital chemical which contribute to the effective functioning of the body. Without a pancreas, many of our most important internal function could not proceed. In the same way that a car needs gas, and break fluid, and engine oil, the body needs all kinds of compounds to martian effective functioning
Digestion really is an incredible process. We tend to think that out mouths do all the work of breaking down food but this is really just the start.
Chewing is just a process of making food small enough to swallow. The real act of digestion doesn't begin to take place until the food reaches the stomach and the intestines. It is then that the vitamins, minerals and other integral elements of food are extracted from the things we eat and absorbed into the different parts of our body.
Reading an article like this never fails to make me marvel at the complexity of the human body. Before I had read this I had never even heard of the trypsin but it sounds like an integral part of the digestive track.
I am also always amazed at how many chemical and chemical processes are going on in the body at any given moment. It is a whole laboratory of activity in there.
Post your comments