At AllTheScience, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What is Absolute Zero?
When a person feels something as hot or cold, what he is feeling is the energy that the object radiates because of motion on a molecular scale. For instance, the molecules in a pot of boiling water are moving much faster than those in an ice cube or a glass of cold water. Physicists theorize that there is a temperature at which molecular motion stops, or is reduced to such a low point that it is unable to transfer any energy that could be considered heat. This theoretical temperature is known as absolute zero.
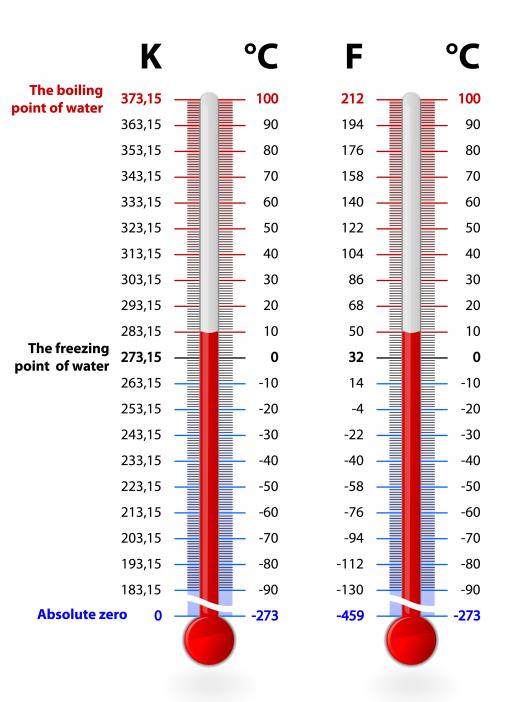
Absolute zero is theoretical because it can never be reached. Scientists, have, however, come very close to producing this temperature in laboratories. The temperature is actually -459.67°F (-273.15°C). On the Kelvin scale, its value is 0°. While this temperature has never quite been reached in a lab or observed in space, scientists have been able to observe the odd behavior and properties of matter that reaches temperatures approaching it.

One of the unexpected results of cooling matter very close to absolute zero was the discovery of a new state of matter. Solid, liquid, and gas are the common states, but when matter, particularly a fluid such as liquid helium, reaches these incredibly low temperatures, it loses all its viscosity and becomes a superfluid. These strange fluids exhibit the ability to flow against gravity, and to a degree, move from their containers into others.
Another phase of matter, called a Bose-Einstein condensate, can also be produced at these extreme low temperatures. Bose-Einstein condensates can only be seen when the temperature of a specimen is brought to within one billionth of 1° of absolute zero, and consequently, only the most specialized laboratories can attempt to study this fragile state of matter. Also, these condensates have so far only been made from microscopically small amounts of matter, on the order of about 10,000 or fewer atoms. They are related to superfluids and behave in somewhat similar ways, but are usually produced from matter in a gaseous state.
The laws of physics that govern Bose-Einstein condensates are not fully understood, and they seem to challenge the things scientists know about the nature of matter. The best way to understand these condensates without an in-depth knowledge of physics is to understand that when matter reaches this point, the atoms in it "collapse" into the lowest possible state of energy, and also begin to behave as if they were no longer discreet particles, but rather waves. Physicists have much more study and research ahead of them in order to fully understand this state of matter, which was only first observed in 1995.
AS FEATURED ON:
AS FEATURED ON:












Discussion Comments
It should be -273 degrees C.... The conversion from C to K is +-273
Post your comments