At AllTheScience, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What Are the Different Types of Dipole?
The term dipole is used in physics and chemistry to describe an electromagnetic phenomenon where two poles, or opposite forces, exist in a given region. A dipole can arise in many different situations, but most dipoles can be categorized as either electric or magnetic. Electric dipoles are often encountered in chemistry in the form of molecular dipole moments — the separation of positive and negative electric charge across a molecule. Magnetic dipoles can be observed in ordinary magnets and compasses, as well as in the micro-scale activity of electrons and other particles.
Electric charge consists of two opposing, or polar, forces: positive and negative charge. These two forces attract each other when brought into proximity, but repulse other charges of like kind — negative repulses negative, for example. The type of charge that a substance has is determined by the distribution of its subatomic particles. Electrons carry a negative charge, while protons, which are found in atomic nuclei, carry positive charge.
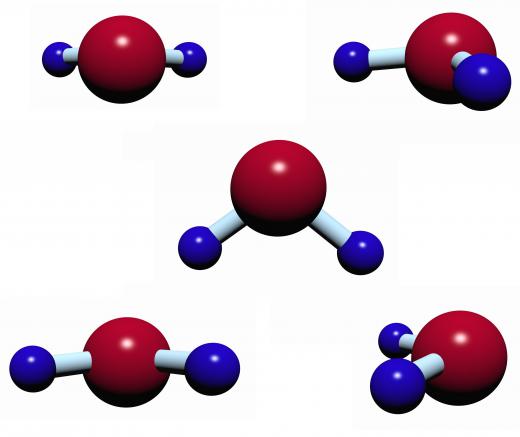
A molecular dipole arises when a molecule has a separation of charge across its structure. For example, a water molecule has a structural arrangement that draws electrons to one side of the molecule, leaving the other side with fewer electrons. A partial positive charge thus forms on one end of the molecule, while a partial negative charge forms on the other, making the molecule polar. In other words, the water molecule has a molecular dipole.

The water molecule’s separation of charge does not change or disappear over time, so it is said to be a permanent dipole. A temporary alignment called an instantaneous dipole can also form in some molecules when electrons temporarily migrate to one part of the structure. Induced dipoles occur when electrons in a molecule are attracted or repulsed by a polar molecule.
Magnetic dipoles make up the second main category of dipoles found in nature. Like electric dipoles, they consist of two opposing poles that attract each other. Bar magnets and compass needles are both examples of this type of system.

The north and south poles of a magnet create a dipole by aligning themselves with a magnetic field. Physicists think of a magnetic dipole as a loop of electric current spinning around an axis as it aligns itself. For example, a compass needle spins until it points north, aligning itself with the earth’s field. The spin of an electron around the nucleus of an atom is also considered a magnetic dipole.
AS FEATURED ON:
AS FEATURED ON:













Discuss this Article
Post your comments