At WiseGEEK, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What is PepZinGI®?
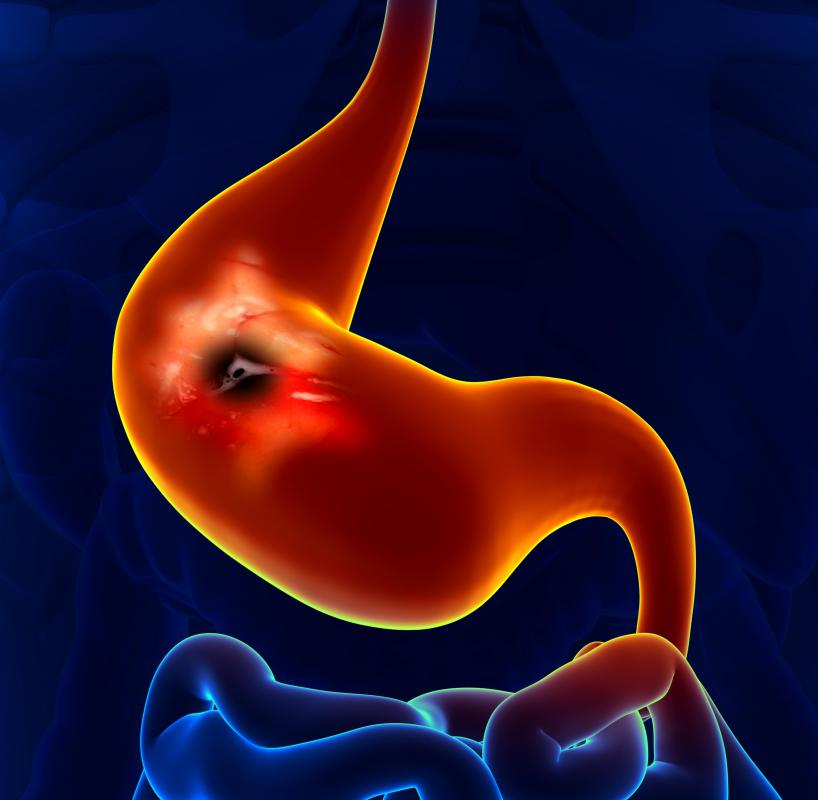
Pepzingi® is the U.S. marketing name for zinc-carnosine, a patented combination of zinc and l-carnosine sold as a health food product or dietary supplement that claims to support digestive and particularly stomach health. It is believed to help heal ulcers, restrain the growth of Helicobacter pylori and relieve the symptoms of indigestion including nausea, belching, diarrhea, heartburn and bloating. Pepzingi® was originally developed in Japan and is typically sold in capsule form in the United States.
Unlike other treatments for stomach and digestion problems, Pepzingi® does not interrupt a digestive process such as acid production. Instead zinc-carnosine supports the body’s natural mucosal defenses that ward off infection and promote healing. Pepzingi® is believed to coat and adhere to the wall of the stomach providing protection for the mucus that shields the stomach lining from the effects of digestive acids.

Additional benefits of Pepzingi® include tempering the growth of the bacteria Helicobacter pylori that is thought to cause the majority of stomach ulcers globally. The supplement also helps inhibit the production of a molecule called interleukin-8 that is linked to inflammation. It also works as an antioxidant that prevents harsh substances like alcohol from damaging the stomach lining.

The U.S. Food and Drug Administration (FDA) recognized and confirmed Pepzingi®'s new dietary ingredient status in 2002. A new dietary ingredient is one that was not sold as a dietary supplement in the United States prior to 1994. Manufacturers who wish to sell a product containing a one must notify the FDA and include documentation that the supplement will be safe if it is consumed according to the recommended use and dosage on the labeling.

There have been multiple randomized and placebo controlled clinical trials and studies of Pepzingi® since it was submitted to the FDA in 1992. In one such study, 299 patients who suffered from gastric upset and discomfort were randomly given either a placebo or Pepzingi® over eight weeks. Of the 139 participants who received doses of Pepzingi®, 92 percent reported that symptoms were moderately improved or better.

There are other potential clinical uses of this supplement beyond the alleviation of stomach discomfort and ulcers. Researchers have explored its possible application as a treatment for osteoporosis, hepatitis, pancreatitis, muscular dystrophy and various taste and smell disorders. It may also be an effective treatment for zinc deficiency.
No serious side effects were linked to Pepzingi® in any of the clinical trials and studies completed by 2010. It does not have any known interactions with any prescription medication or other dietary supplements. A medical practitioner should be consulted prior to starting a regimen of this supplement in order to avoid any unforeseen complications.
AS FEATURED ON:
AS FEATURED ON:













Discuss this Article
Post your comments