At TheHealthBoard, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What is Nesidioblastosis?
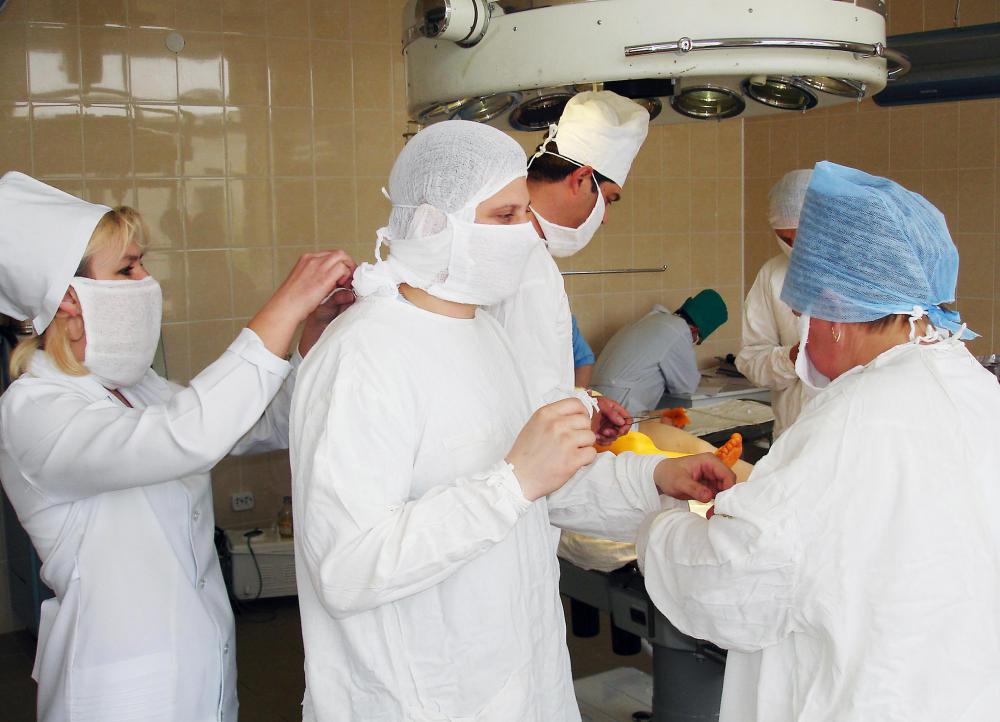
Nesidioblastosis is dangerous persistent hypoglycemia, or low blood glucose levels, that is caused by too much insulin, a condition called hyperinsulinism when it is not caused by an insulin-producing tumor. Hypoglycemia is a potentially dangerous condition that can cause symptoms such as headache, confusion, shakiness, blurred vision and heart palpitations. It also can cause seizures, brain damage, coma or even death. Infants with this condition need surgery to remove part of the pancreas — called a partial pancreatectomy — or they can suffer severe brain damage or death.
Scientist George F. Laidlaw coined the term "nesidioblastosis" in 1938 to refer to babies with abnormalities of the pancreas, including too many pancreatic beta cells, beta cells where they should not be and larger-than-normal beta cells. The term "nesidioblastosis" fell out of favor when scientists discovered that some people have the physical features of nesidioblastosis without developing hypoglycemia, so doctors began identifying disorders by their symptoms instead, such as persistent hyperinsulinemic hypoglycemia of infancy (PHHI) or congenital hyperinsulinism. Scientists in the 1980s discovered that many patients with hyperinsulinism have lower levels of somatostatin, a hormone that inhibits the release of insulin, so hyperinsulinism involves more than just abnormal beta cells. Although persistent hypoglycemia caused by hyperinsulinism usually occurs in babies age 2 or younger, when the pancreas is still developing, physicians began using the term "nesidioblastosis" in the early 21st century to refer to persistent hyperinsulinemic hypoglycemia in adults who develop beta cell abnormalities following gastric bypass surgery.

In the 1930s, physicians noticed that some babies who developed severe persistent hypoglycemia had very high levels of insulin in their blood. Pathologists discovered that many of these babies had specific pancreatic abnormalities involving beta cells, which are insulin-producing cells located in the islets of Langerhans of the pancreas. Babies with severe hypoglycemia because of excess insulin production had enlarged beta cells and beta cells where there should be none, such as in and around the pancreatic ducts.

Scientists in the 1970s and '80s discovered that some people have the pancreatic abnormalities seen in nesidioblastosis without experiencing persistent hypoglycemia or hyperinsulinemia. Pathologists have found abnormal formation and placement of beta cells in people with cystic fibrosis, Zollinger-Ellison syndrome or sudden infant death syndrome and even in healthy infants. Hyperinsulinism is not usually caused by beta cell problems alone but by a combination of issues, including too few pancreatic delta cells, which release somatostatin, a hormone that inhibits the release of insulin. These discoveries caused the name "nesidioblastosis" to fall out of favor with many doctors, so that "PHHI" "congenital hyperinsulinism," "nesidiodysplasia" and "islet cell dysmaturation syndrome" became the preferred terms.

Although nesidioblastosis almost always occurs in babies age 2 or younger, rare cases can occur in adults, sometimes as a side effect of gastric bypass surgery. As of early 2011, the cause of adult-onset nesidioblastosis remained unclear. Some adults with nesidioblastosis have antibodies against beta cells in their blood serum, which might cause beta cells to grow too much as a reaction to being attacked by the body's immune system; other adult nesidioblastosis patients have high levels of some beta cell growth factors and higher numbers of receptors for beta cell growth factors in the pancreas, which might be causing the beta cells to grow abnormally. Both of these conditions have been found in gastric bypass patients who develop nesidioblastosis. Nesidioblastosis is a rare complication of gastric bypass surgery, the serious nature of the disorder and the growing number of gastric bypass procedures mean that physicians need to educate postoperative patients on the symptoms of hypoglycemia and screen hypoglycemic patients for nesidioblastosis.
AS FEATURED ON:
AS FEATURED ON:

















Discussion Comments
My name is Mario and I still have that nesidioblastosis and neogenese on me. The Glp-1 is secreted by my L-cell and that affects all my system. If someone wants to talk about nesidioblastosis after RYGB, we can talk about it. I've studied that problem for the last four years.
Post your comments