At WiseGEEK, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What is an Amyloid Precursor Protein?
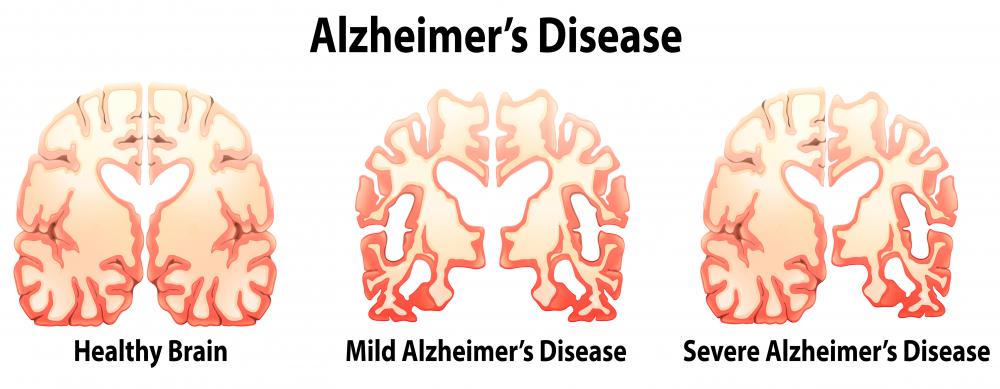
An amyloid precursor protein, also known as APP, is involved in biochemical processing in the brain that is thought to lead to Alzheimer’s disease. This protein is processed in several manners. Some types of processing lead to the production of clumps of fibers in the brain known as amyloid plaques. These build up and form tangles that are thought to kill neurons and interfere with brain functions.
This protein is widespread throughout the body and is found in nerve synapses. Its normal function is unknown. It has been widely studied because its processing has been implicated in the development of Alzheimer’s disease. Amyloid precursor protein is an integral membrane protein. It is embedded in membranes within the cell, with part of the protein protruding above the membrane and a smaller piece found below.

Proteins are long chains of amino acids, which can be cut by protease enzymes to form smaller chains called peptides. Many proteases, such as those involved in digestion, can act on a broad range of proteins. Others, however, are very specific and only cleave particular proteins. Amyloid precursor proteins are cleaved by several types of proteases, known as secretases. These types of proteases cleave off pieces of proteins that are located in cellular membranes.

There are three types of secretases that act on amyloid precursor protein — alpha, beta, and gamma secretase. There is no abnormality associated with the activity of alpha secretase. The sequential activities of beta and gamma secretase, however, lead to the production of peptides known as beta-amyloid. These peptides range from 39 to 42 amino acids in length. They form aggregates known as beta-amyloid plaques. These are found in the brains of patients with Alzheimer's and some other brain disorders.

The cause of Alzheimer’s disease has not been definitely proven. The current model, however, postulates that these beta-amyloid plaques are responsible for the symptoms of the disorder. Genetic studies indicate that amyloid precursor protein plays a role in the development of Alzheimer’s disease.
The gene for this protein is highly complex and is also known as APP. Many mutations in this gene result in early-onset Alzheimer’s disease. Over 25 such mutations have been identified. They have been found to be responsible for 10-15% of all of the cases of Alzheimer’s diseases that strike early in life. About 5-10% of all Alzheimer’s cases are due to inherited susceptibility.

Efforts are underway to block the processing of amyloid precursor protein that leads to beta-amyloid peptides, by limiting the activities of beta and gamma secretase. One beta-amyloid in particular is more likely to form toxic fibers. This is a 42-amino-acid long peptide that is stickier than the shorter peptides. Researchers have developed a drug that acts to favor gamma secretase’s production of shorter and less toxic peptides, in the hope of reducing beta-amyloid plaques, and thus limiting the progression of Alzheimer’s disease.
AS FEATURED ON:
AS FEATURED ON:














Discuss this Article
Post your comments