At WiseGEEK, we're committed to delivering accurate, trustworthy information. Our expert-authored content is rigorously fact-checked and sourced from credible authorities. Discover how we uphold the highest standards in providing you with reliable knowledge.
What Are Intravenous Solutions?
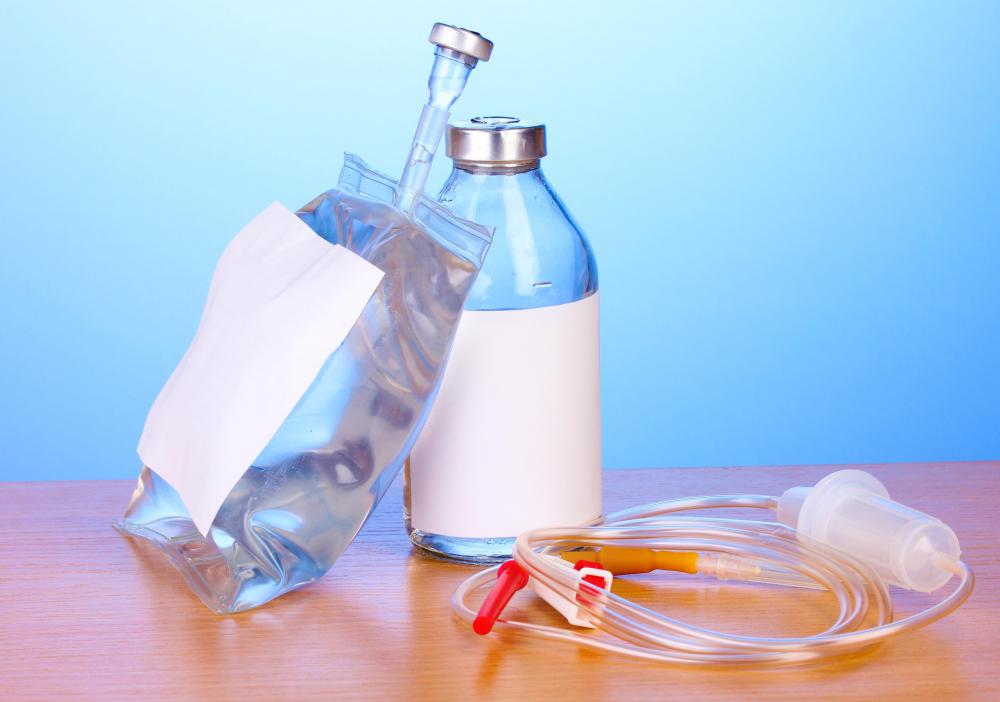
Intravenous solutions are medications delivered through intravenous (IV) therapy. A number of solutions exist, from simple saline to expensive chemotherapy medication. No matter the medication, using intravenous solutions can cause certain complications in patients.
One of the simplest and most commonly used intravenous solutions is saline. Medical saline is a solution of water and salt that has the same salinity as human blood. It has three uses; the first is to raise blood pressure where needed. The second is to act as a solvent for other medication. The third is to replenish fluids when a patient cannot drink water.

Another common medication delivered in intravenous solutions is antibiotics. In many cases, the infection has progressed to a point that requires hospitalization. Delivery through intravenous solution allows a constant infusion of antibiotic medication, an advantage when compared to oral antibiotics where effectiveness varies throughout the course of the day. This difference can be life-saving in cases where oral antibiotics have failed to halt an infection.

For individuals who experience blood loss as the result of an accident or another event, a blood transfusion is a necessary treatment to save a patient's life. Blood transfusions are a necessity during many surgical procedures as well. It is possible to extend the use of donated blood by mixing it with saline.
Intravenous sodium bicarbonate is an intravenous solution used to treat acidosis of the blood. Acidosis, or raised acidity, has a number of causes, from cancer to drug abuse. Though sodium bicarbonate solution does not solve the underlying problem, it stops the acidosis from causing harm to the body's tissues. Throughout treatment, it is necessary for medical professionals to ensure that the blood does not become too basic in the process of treatment.

Chemotherapy is a category of intravenous solutions used to treat various cancers. These medications, most synthetically produced after years of laboratory research, are dissolved into saline during preparation. As too little medication will have no effect on a cancer and too much might irrevocably harm the patient, a precise mixture is necessary.
There is some risk to patients when delivering any intravenous solution. During any medical procedure that requires cutting or breaking the skin for prolonged period of time, infection is a possibility. With therapies such as blood and saline, fluid overload can occur; this event is when the body cannot process an increased amount of fluid. Finally, a rarer complication is hypothermia due to the infusion of cool or cold intravenous solution; this condition can become life-threatening if it causes heart fibrillation.
AS FEATURED ON:
AS FEATURED ON:












Discuss this Article
Post your comments